Entropy is one of the factors that determines the free energy of the system. 
Quantitatively, entropy, symbolized by S, is defined by the differential quantity d S = δ Q / T, where δQ is the amount of heat absorbed in a reversible process in which the system goes from one state to another, and T is the absolute temperature. Entropy is an extensive state function that accounts for the effects of irreversibility in thermodynamic systems. In recent years, entropy has been interpreted in terms of the " dispersal" of energy. Entropy change has often been defined as a change to a more disordered state at a microscopic level. In contrast the first law of thermodynamics deals with the concept of energy, which is conserved. Spontaneous changes occur with an increase in entropy. The concept of entropy in thermodynamics is central to the second law of thermodynamics, which deals with physical processes and whether they occur spontaneously. It's a fascinating and complex subject which really can't be summarised in one post.Ice melting - classic example of entropy increasing described in 1862 by Rudolf Clausius as an increase in the disgregation of the molecules of the body of ice. A vector with relatively 'high' entropy is a vector with relatively high information content. 
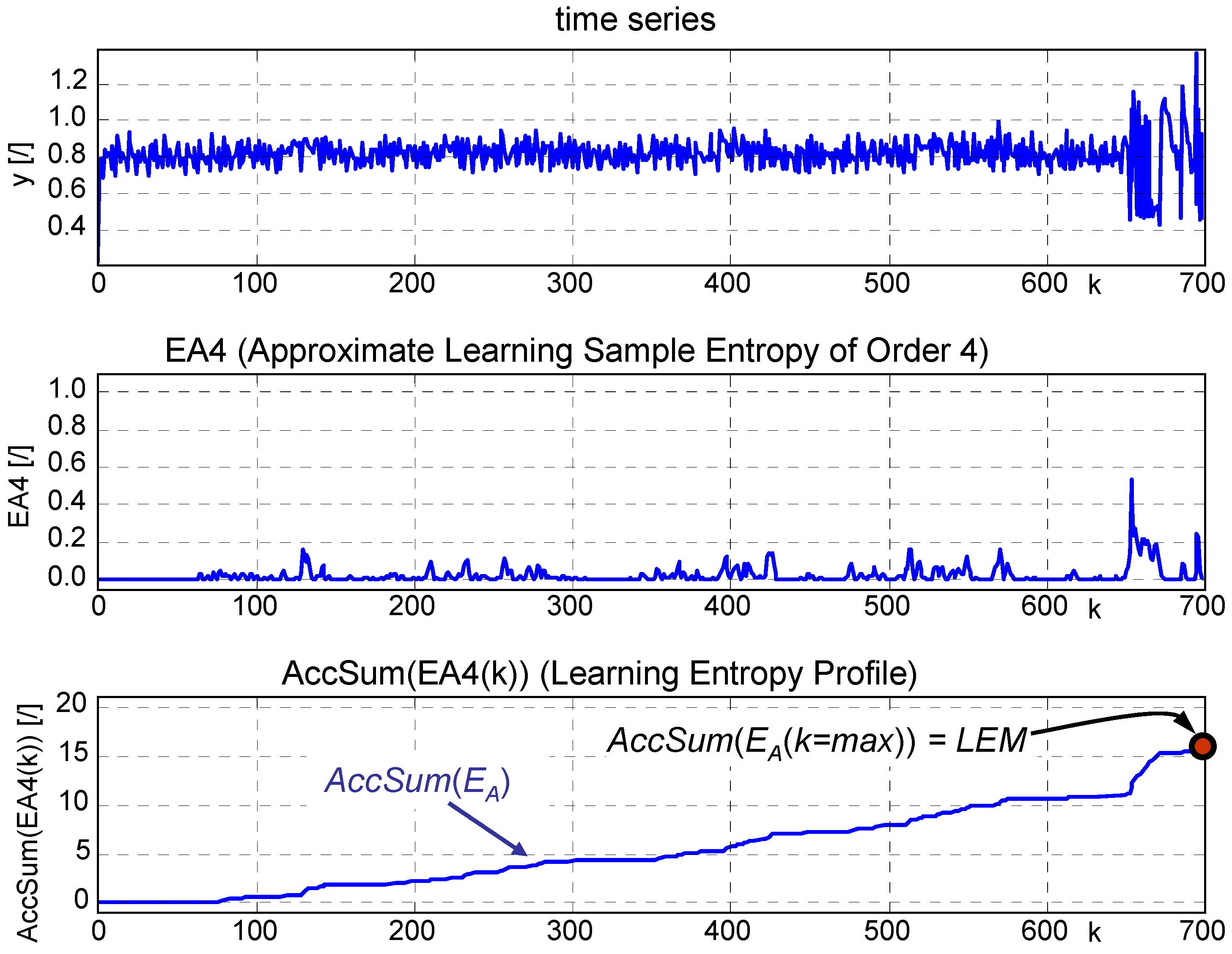
A vector with relatively 'low' entropy is a vector with relatively low information content. A component with low entropy is more homogenous than a component with high entropy, which they use in combination with the smoothness criterion to classify the components.Īnother way of looking at entropy is to view it as the measure of information content. In the context of the paper low entropy (H(s_m) means low disorder, low variance within the component m. So what does this mean? In image processing entropy might be used to classify textures, a certain texture might have a certain entropy as certain patterns repeat themselves in approximately certain ways. The probability density p_n is calculated using the gray level histogram, that is the reason why the sum runs from 1 to 256. Here is the probability that outcome s_m happens. H(s_m) is the entropy of the random variable s_m. As the level of disorder rises, the entropy rises and events become less predictable.īack to the definition of entropy in the paper: 
Entropy can serve as a measure of 'disorder'. One way to view entropy is to relate it to the amount of uncertainty about an event associated with a given probability distribution. They are talking about Shannon's entropy. The target component is a tumor and the paper reads: "the tumor related component with "almost" constant values is expected to have the lowest value of entropy."īut what does low entropy mean in this context? What does each bin represent? What does a vector with low entropy look like? But I'm failing to understand what entropy is in this case.Īnd they say that '' are probabilities associated with the bins of the histogram of '' In the paper I'm reading, the authors wish to select a component m for which matches certain smoothness and entropy criteria. T is the total number of pixels in the image, is the value of the source component (/signal/object) i at pixel j The output of the algorithm is a matrix, which represents a segmentation of an image into M components. I'm reading an image segmentation paper in which the problem is approached using the paradigm "signal separation", the idea that a signal (in this case, an image) is composed of several signals (objects in the image) as well as noise, and the task is to separate out the signals (segment the image). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
